
Carrier screening, also referred to as preconception genetic screening, helps individuals and reproductive partners understand their risk of passing along autosomal recessive and X-linked genetic conditions to their children. This method is being increasingly adopted as a preventive, population-level health intervention that specifically benefits couple’s or individuals in the family planning stage. With the rise of Next-Generation Sequencing (NGS), genetic screening has expanded from a small number of ethnicity-based tests to broad, pan-ethnic panels covering hundreds of conditions. At Golden Helix, we’ve built VarSeq to help clinical laboratories implement carrier screening workflows at scale. Users can automate the most labor-intensive steps of tertiary analysis so their labs can meet growing demand efficiently and consistently.
The Challenge of Scaling Expanded Screening
Carrier screening targets conditions that are often clinically silent in parents, severe or resulting in poor quality of life in affected children, and frequently preventable or manageable if risk is identified early. This screening method has already changed disease incidence in real populations by giving the gift of informed choice. Community-based screening programs initiated in the 1970s led to a >90% reduction in Tay-Sachs incidence in high-risk populations through informed reproductive planning, as reviewed in this article. Yet, adoption of this test remains far below its theoretical reach, as most screenings are still performed during pregnancy, not preconception, and many couples of reproductive age are never offered screening at all.
VarSeq helps to reduce the gap between clinical value and real-world utilization, thereby presenting both a public-health opportunity and an operational win for laboratories. Expanded screening introduces complexity in these ways:
- Large number of genes and conditions to analyze
- Partner-aware interpretation requirements
- Recessive and X-linked inheritance logic that needs to be applied
- There is a need for clear, consistent reproductive risk reporting
Without automation, screening becomes labor-intensive, difficult to scale and prone to inconsistency across cases and analysts. This is where VarSeq’s tertiary analysis capabilities become critical. VarSeq serves as the automation backbone for screening workflows, handling the complexity of partnered analysis while maintaining clinical rigor.
With VarSeq genomics, labs can:
Import and Pair Reproductive Partners
Carrier screening analysis begins by importing samples and explicitly pairing reproductive partners. VarSeq’s partner-aware project structure ensures that all downstream analysis is performed in the correct biological and clinical context. This pairing enables VarSeq to move beyond single-sample variant lists to couple-level risk assessment.
Detect and Prioritize Carrier Variants
VarSeq applies standardized quality, frequency, and annotation filters to identify variants relevant to carrier screening. Built-in ACMG-based scoring helps prioritize variants most likely to be clinically significant, creating a reproducible foundation for interpretation.
Identify Shared Carrier Status and Reproductive Risk
A defining feature of genetic screening is evaluating shared genetic risk across partners. VarSeq’s shared carrier gene detection logic identifies genes in which both partners carry relevant variants and flags them for review.
Apply Best Practices
Best-practice analysis for carrier screening distinguishes between potential compound heterozygous risk (different variants in the same recessive gene) and potential homozygous recessive risk (same variant in both partners) and X-linked carrier status with sex-specific reproductive implications. By performing this analysis at the gene level, VarSeq ensures that clinically meaningful risks are not missed.
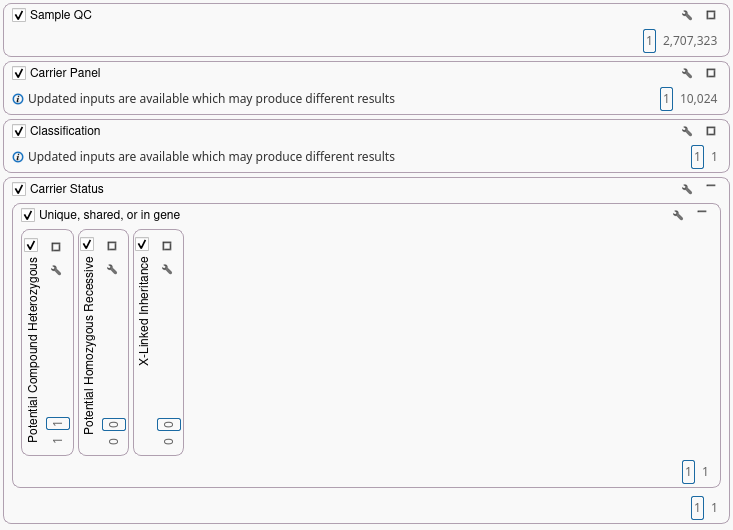
Use ACMG or Lab-Curated Carrier Gene Lists
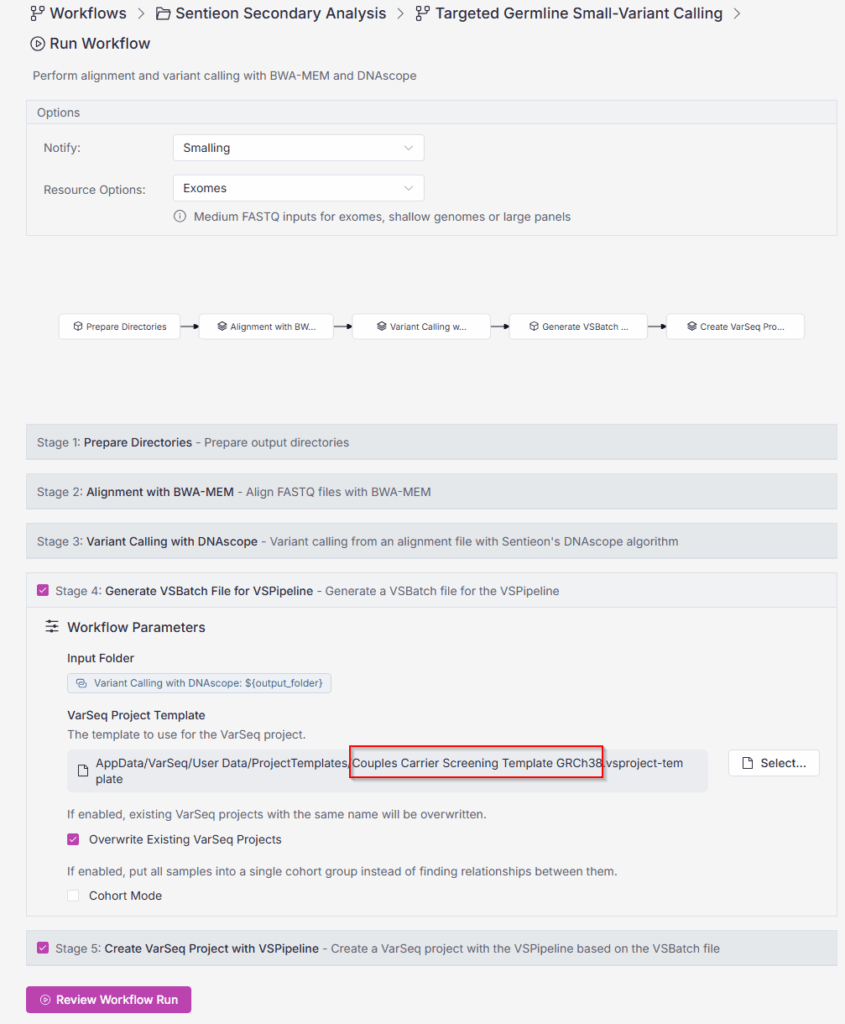
VarSeq supports both ACMG-recommended carrier gene sets, providing a standardized baseline and lab-curated or expanded panels, reflecting institutional preferences, population needs, or commercial offerings. Gene lists can be versioned and tied to validated project templates which is an essential best practice for consistency, traceability, and regulatory compliance.
Integrate Screening into Broader Germline Workflows
Carrier screening does not need to exist in isolation. VarSeq’s flexible filtering and tagging system allows screening logic to be embedded within larger germline analysis projects, enabling laboratories to reuse infrastructure, reduce pipeline sprawl and scale efficiently as test menus expand. Furthermore, VarSeq Warehouse 3.0, our integrated platform enables users to go from FASTQ all the way to finished projects in a single workflow and a single environment.
Conclusion
By automating tertiary analysis in carrier screening, leveraging partner pairing, shared carrier detection, inheritance-aware filtering, and structured outputs, VarSeq enables laboratories to scale screening programs without sacrificing quality or consistency. As genetic screening continues to move toward standard-of-care, we provide efficient and automated analysis that will facilitate labs’ increasing adoption of this test, which will drive the realization of its full public-health impact.
If you would like to learn more about implementing couples carrier screening workflows using VarSeq, please reach out to our team today.



