
Over the past several months, we’ve highlighted the powerful new capabilities introduced with VarSeq Warehouse 3.0. Today, I’d like to focus on a familiar feature that’s been thoughtfully enhanced, assessment catalog record management. This update significantly improves data quality, role-based collaboration, and regulatory compliance.
Variant assessment catalogs have always been a core component of the VarSeq platform, allowing users to save and store single nucleotide variants (SNVs) and copy number variants (CNVs). With VarSeq Warehouse 3.0, this functionality has been expanded to include structural variants and sample-level assessments as well. More importantly, these records can now be managed through a permission-based workflow, where specific users have designated roles to create, review, and publish variant interpretations.
In this post, I’ll walk through what this updated lifecycle management process looks like and explain why it’s a game-changer for clinical labs striving for quality, consistency, and compliance in their genomic workflows.
Variant and Sample Record Management in VS Warehouse 3
In VS Warehouse 3, assessment catalog records can easily be queried, created, edited, and removed. With great power comes great responsibility, so what we have done is allow users control over the entire lifecycle of creating assessments to finalized and published assessments.
The first step is to configure your assessment catalog with the appropriate lifecycle “gates” which include draft, review, and publish stages. You can choose to implement just one, two, or all three gates depending on the level of control your workflow requires.
For example, a rigorously maintained catalog used to track well-established pathogenic variants may benefit from all three stages to ensure careful review before publication. On the other hand, more flexible catalogs can opt for a simplified workflow with fewer checkpoints.
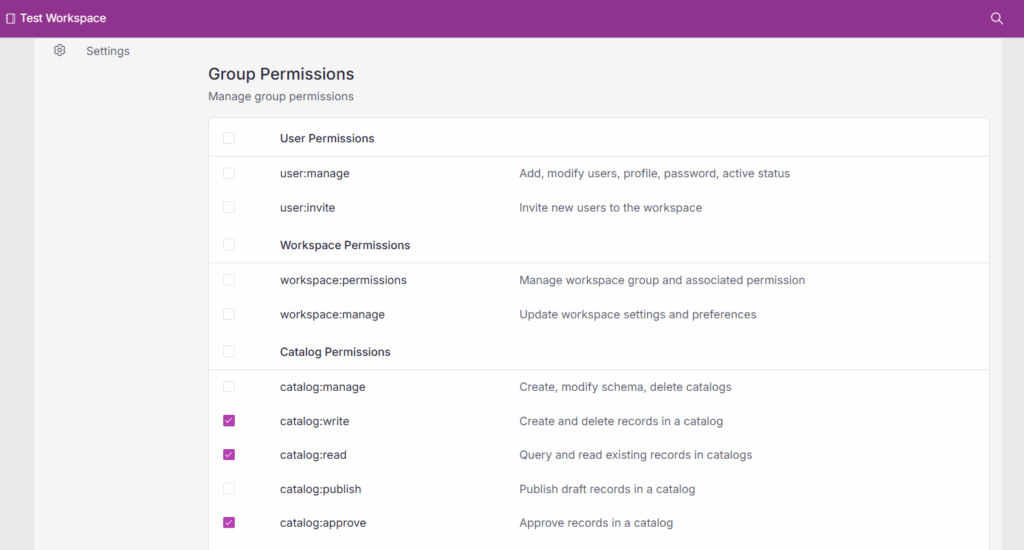
Once gates are configured, role-based permissions can be assigned to users or groups for each stage of the lifecycle. This is essential for creating a clear division of responsibilities and maintaining an auditable change history.
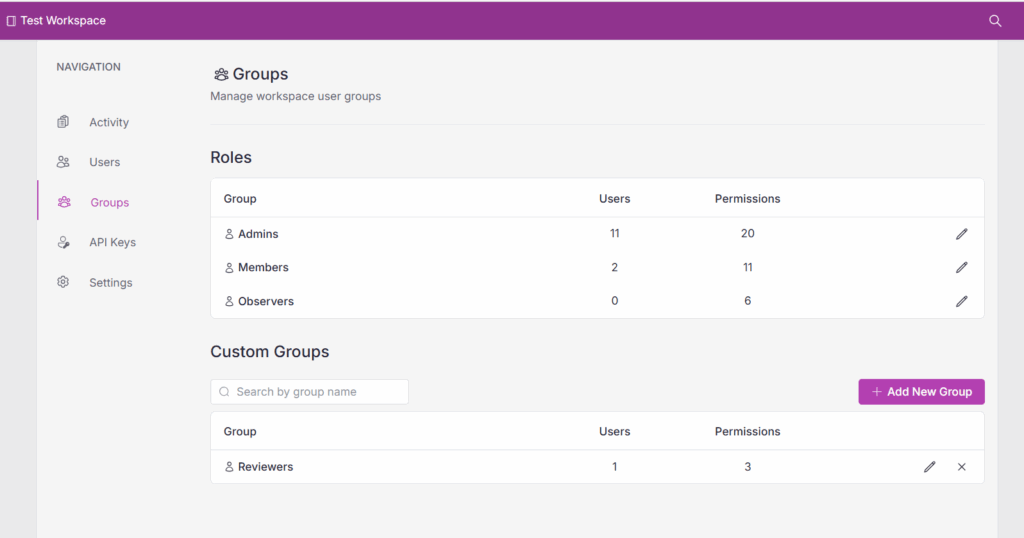
Workspace admins can manage default groups like Admins, Members, and Observers, or define new custom groups such as Reviewers with tailored permissions (Figures 1).
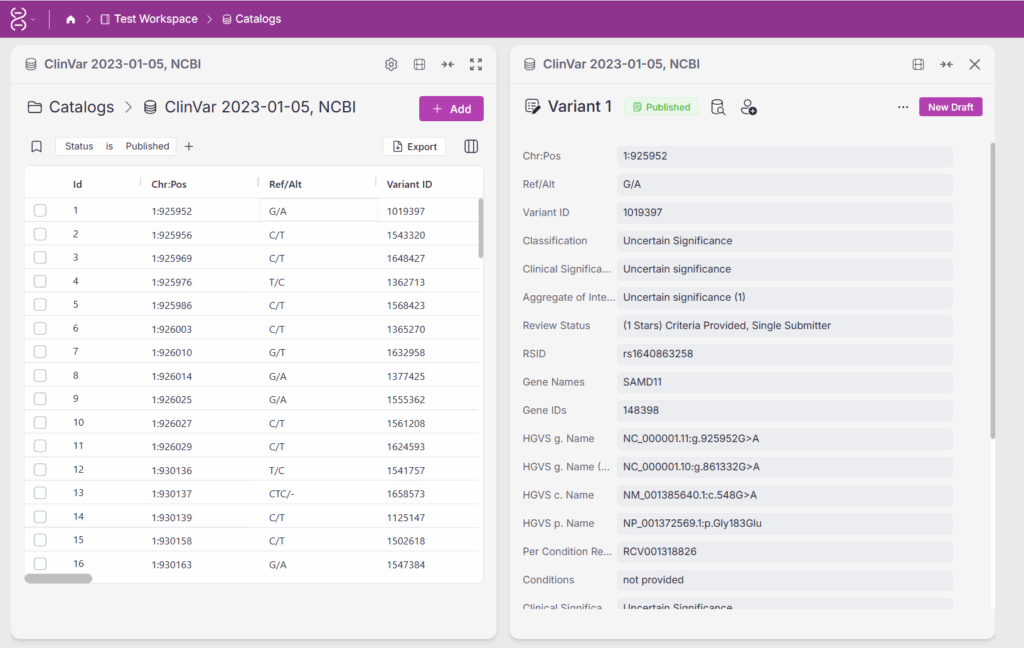
With permissions set, the record management process begins. In Figure 2, we see a variant assessment catalog. On the left is the list of variant records; on the right is a selected record currently in the published state. In the top-right corner, users with the appropriate ‘write’ permissions can create a new draft.
In Figure 3, a variant record is shown in the draft state, and its Classification field has been edited. Users with draft permissions can view and further edit this record. For more control, the draft can be assigned to a specific user or group, ensuring focused ownership and reducing ambiguity.
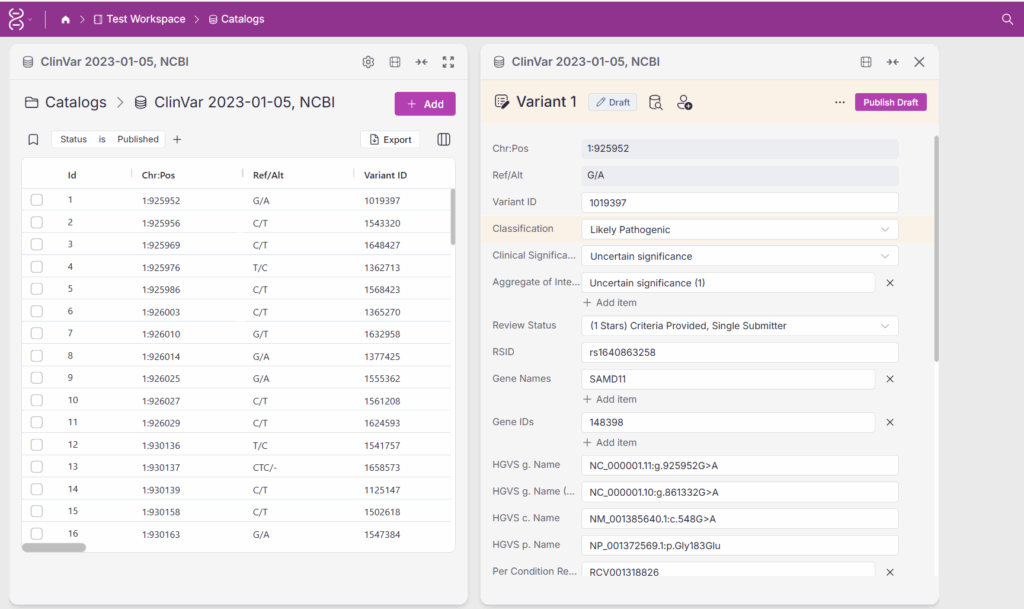
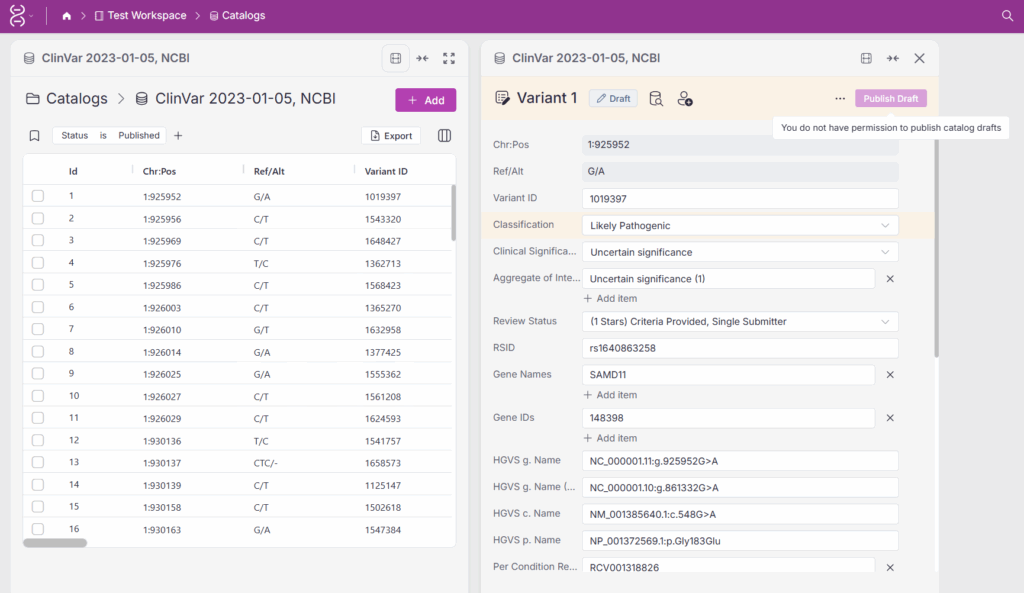
Once a draft is finalized, it can move to the review stage and ultimately to publication. In Figure 4, the variant record is ready to be published. It has already been evaluated by three users, reinforcing the rigor and reliability of the analysis. However, the current user does not have permission to publish, highlighting the importance of controlled publishing permissions.
Benefits of the Record Lifecycle Management Process
1. Ensures accuracy, accountability and confidence for variant assessments
Implementing multiple review stages in the variant curation process significantly reduces the likelihood of errors in interpretation or clinical annotation. This structured approach ensures that only vetted, high-confidence genomic data progresses to the published state. Each change made to a record is traceable, providing full visibility into who made what edits and when, which supports accountability and auditability.
By the time a record is published, it has undergone thorough review and approval, instilling confidence in its accuracy among downstream users such as clinicians and researchers. This process minimizes the risk of misinterpretation or reliance on unverified findings and ultimately helps prevent clinical misguidance resulting from erroneous variant classifications.
2. Provides structured, role-based collaboration
Establishing defined roles—such as curator, reviewer, and publisher—creates a clear separation of responsibilities within the variant curation workflow. This structure enables effective collaboration among subject-matter experts, bioinformaticians, and clinical leads, ensuring that each contributor operates within their area of expertise without overlap or conflict. It also encourages a robust peer review process, which is essential in the clinical genomics world. Additionally, this role-based framework supports auditability by allowing for retrospective reviews, investigations, and quality control, all of which are vital for maintaining the integrity and reliability of genomic data.
3. Supports clinical compliance, risk mitigation, and traceability
A structured, permission-based record management system aligns with regulatory expectations such as CLIA, CAP, and ISO standards by supporting robust auditability and clearly defined approval chains. It also helps fulfill documentation and version control requirements essential for clinical variant curation. By ensuring that only reviewed and approved data can be published, the system mitigates the risk of unverified or potentially harmful information reaching end users. Furthermore, it promotes accountability by maintaining a complete audit trail of the variant record’s history, including who proposed, reviewed, and ultimately approved each change.
For more information about how to set up assessment catalog record lifecycle management with VS Warehouse 3 please do not hesitate to reach out to our support team!



