How VarSeq Helps Explore the Therapeutic Landscape for CRC Patients

Colorectal Cancer Awareness Month for CRC Patients
In honor of Colorectal Cancer Awareness Month, we’re taking a close look at how VarSeq’s VSClinical AMP workflow and evidence from Genomenon and Golden Helix CancerKB can expose the right treatment strategies for CRC patients, including those who might otherwise be overlooked.
Colorectal cancer is one of the most common and deadly malignancies in the world, with approximately 1.9 million new cases and over 900,000 deaths every year. Even though there has been progress in chemotherapy and targeted therapy, treatment options for many CRC patients, especially those with advanced disease still remains limited. Immunotherapy has made a huge difference in treating solid tumors across the board, but CRC is an exception to the rule.
Immunotherapy in Colorectal Cancer
Roughly 15% of CRCs have defects in the DNA mismatch repair system- typically mutations in the MMR genes. When MRR is deficient, microsatellites accumulate and produce microsatellite instability. The good news about tumors that are microsatellite instability high is that they are recognizable by the immune system and thus respond well to immune checkpoint inhibitor (ICI) therapy. The remaining 85% of CRC patients that are microsatellite stable and MRR-proficient are not candidates for ICI therapy, highlighting the importance to test and report MSI/MMR status for CRC patients.
VarSeq Use Cases
Example 1
TMB- Ultrahypermutated
- The FDA has approved pembrolizumab for TMB-high solid tumors, defined at a threshold of ≥10 mutations per megabase (mut/Mb
- In CRC, the ultrahypermutated tier of TMB defined as >100 mut/Mb is where the most dramatic immunotherapy responses are observed.
- TMB ultrahypermutated tumors display a pre-existing inflamed microenvironment and exceptional sensitivity to ICI therapy that likely exceeds what their TMB number alone would predict.
POLE LoF mutations in codons 268-471
- POLE encodes the proofreading exonuclease subunit of DNA polymerase epsilon, the enzyme responsible for the real-time error correction that occurs during DNA replication. This process occurs before the MMR system even comes into play.
- LoF mutations result in replication errors that overwhelm all downstream repair, generating mutational burdens that can reach hundreds to thousands of mutations per megabase.
- Many POLE-mutant CRC tumors test as microsatellite stable, but the tumor is actually one of the most immunologically active in oncology!
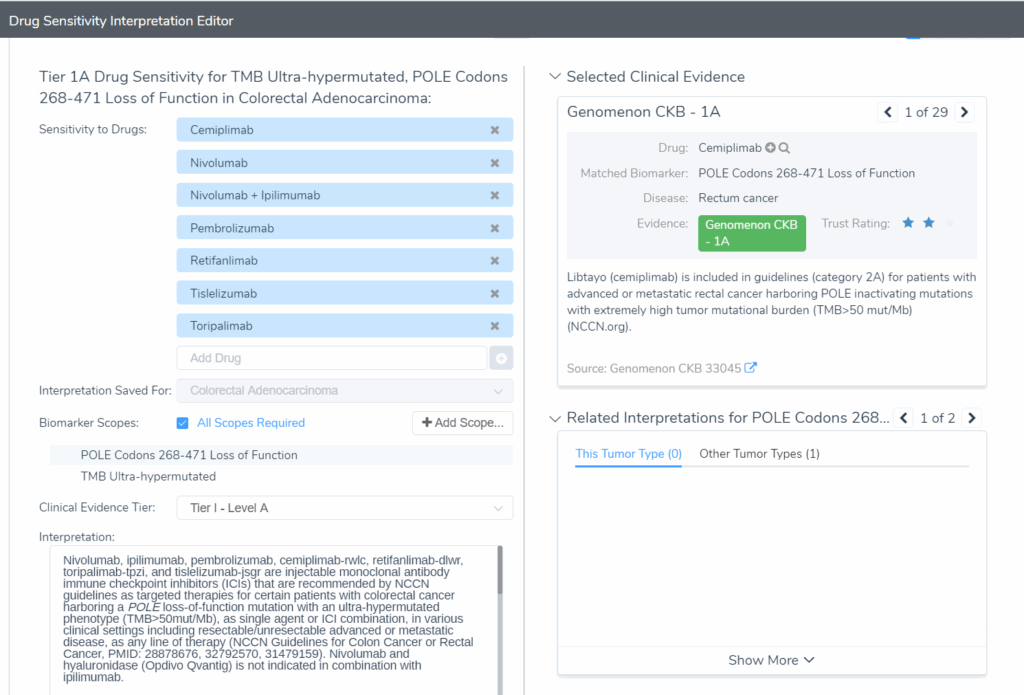
VarSeq surfaces this distinction within the VSClinical AMP workflow, helping teams contextualize TMB quantification alongside the variant-level findings that explain why a tumor has reached that level of hypermutation.
In the VarSeq use case below, a patient presents with an MSS result but VarSeq’s VSClinical AMP workflow identifies a POLE exonuclease domain mutation alongside an ultrahypermutated TMB status. The CancerKB evidence panel surfaces multiple treatment strategies with corresponding interpretations, ensuring the complete molecular picture reaches the clinician and the patient is not erroneously excluded from ICI therapy.
Example 2: The Other 85% of Colorectal Cancer Cases
Those CRC patients with metastatic disease who are microsatellite stable, MMR Proficient, and POLE/POLD1 wild type who currently do not have immunotherapy options, until potentially now.
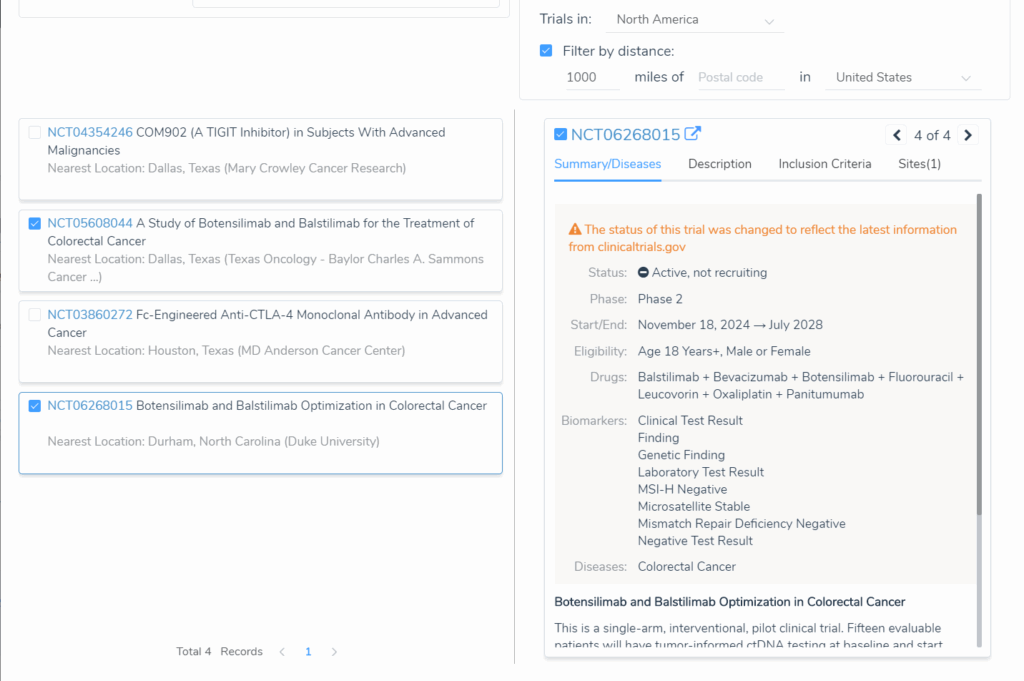
Currently there are active clinical trials for the combination therapy Botensilimab + Balstillmab. Botensilimab is a next-generation anti-CTLA-4 antibody engineered with an Fc region that enhances immune effector function that goes beyond simple checkpoint blockade to actively stimulate and sustain anti-tumor T cell responses. Balstilimab is a conventional anti-PD-1 checkpoint inhibitor. Together, they target both the CTLA-4 and PD-1 axes simultaneously, attempting to both prime and maintain an immune response in tumors that have historically been resistant to either approach alone.
VarSeq has this population covered too. Even for patients who are MSS, TMB-low, and POLE/POLD1 wild-type, the VSClinical AMP workflow draws on clinical trial annotations shows the active BOT/BAL trial along side the clinical context clinicians need to assess eligibility. For a patient who has exhausted standard therapies, the difference between a report that says “no actionable findings” and one that identifies an active Phase 2 trial could be the difference between no options and a meaningful chance at durable response.
VarSeq’s integration of POLE and POLD1 loss-of-function variant classification, TMB ultrahypermutation stratification, MSI status, and Genomenon and CancerKB clinical evidence annotation gives molecular tumor boards the comprehensive, layered view they need in a single workflow, with interpretable evidence at every step.



