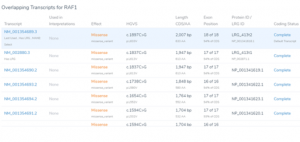
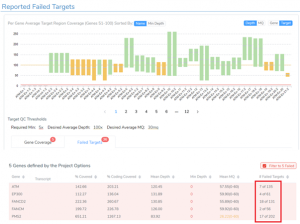
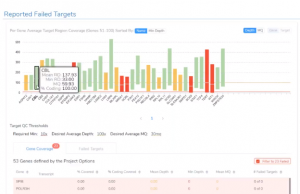
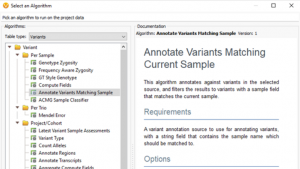
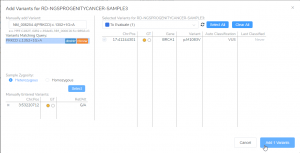
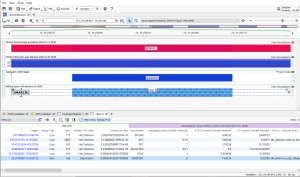
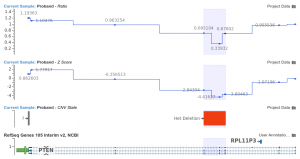
Featured in The Journal of Precision Medicine: Golden Helix’s Recent Release for a New Workflow for Interpretating and Reporting Copy Number Variants in Concordance with the Recently Updated ACMG Guidelines: Introduction

Have you seen us in The Journal of Precision Medicine? Last week, our team released VarSeq 2.2.2 loaded with a number of updates and VSClinical’s highly-anticipated ACMG-CNV Guideline workflow! We have spent the past several months sharing webcasts and blogs on this new capability. We are honored to also have…
Read more →