Introducing Drugs and Trials for Cancer Diagnostics

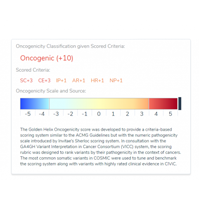
When interpreting a variant using the AMP guidelines for somatic variant interpretation, clinicians must determine whether the variant can be considered a biomarker that affects clinical care by predicting sensitivity, resistance, or toxicity to a specific therapy. Such a determination requires the investigation of multiple evidence sources, including clinical trials,…
Read more →