Introducing VarSeq 3.0.0: Faster, Smarter, Fully Integrated with VSWarehouse 3

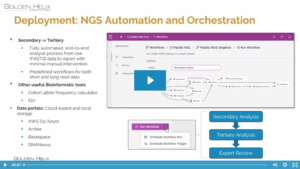
VarSeq 3.0.0 is officially here, and it’s one of our most significant releases yet! The headline feature of this update is full integration with VSWarehouse 3, including a new login system that supports both single-user mode and server-managed authentication. Users can now choose to work locally or seamlessly store projects…
Read more →