In the last year, we have seen a surge in customers moving to whole genome sequencing. Not only does whole genome sequencing provide unparalleled gene coverage compared to whole exome, but depending on the kind of sequencing, you may expect to see additional file types as well. For example, our partners at PacBio will provide an additional VCF containing copy… Read more »
Golden Helix has a long history of developing a high-quality NGS-based CNV caller. This has become even more apparent with our latest publication, where we teamed up with Twist Bioscience to demonstrate VarSeq capability of calling large copy number variants utilizing their clinical whole exome sequencing data. You can access the recent publication here: Analyzing Performance of Twist Bioscience Exome… Read more »
The recent release of VarSeq 2.6.0 was filled with so many customer-requested features (for example, our long-awaited PGx workflow!) that some of our other new features have not yet had their time in the spotlight. For this blog, we are thrilled to announce that with the release of VarSeq 2.6.0, we have made WGS CNV calling more user-friendly than ever… Read more »
In this blog post, we will explore the nuances of variant calling and import in the context of VSPGx. We will discuss the importance of integrating must-call variant definitions into the calling processes and provide guidance on incorporating copy number variants (CNVs) and structural variations (SVs) into your PGx analysis workflow. Must Call Variant Files To ensure the optimal performance… Read more »
In the rapidly evolving field of genomics, innovative technologies, and methodologies are constantly being developed to enhance our understanding and diagnosis of genetic disorders. Golden Helix’s VarSeq and VS-CNV software platforms stand at the forefront of this revolution, offering powerful tools for genetic data analysis and interpretation. This blog highlights recent research endeavors where these tools have been instrumental. From… Read more »
Recently, at ACMG 2024, we unveiled a collaborative research project with Twist Biosciences. By utilizing Twist Bioscience enhanced Exome 2.0 Plus Comprehensive Exome Spike-In capture panel with added backbone probes, we developed a multi-modal CNV caller designed specifically for target-capture NGS data to detect single-exon to whole-chromosome aneuploid CNV events. We are proud to say that from Golden Helix, Andreas… Read more »
Every month we compile customer publications that reference us, and every time I am excited to see the amazing work being done around the world. From pediatric heart conditions to rare diseases, or a Thai clinical study on dilated cardiomyopathy, it is always a pleasure to see the Golden Helix Software suite helping research and clinical genetics. Below are a… Read more »
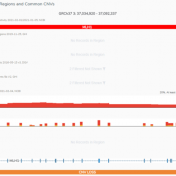
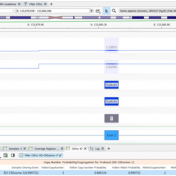
The ability to analyze copy number variants (CNVs) is an important aspect of any clinical or research workflow. While calling CNVs can be a challenging engineering problem, we are thrilled by our capacity to detect, analyze, and catalog CNVs all in the same place with VarSeq-CNV. In this blog, we will dive into the particulars of detecting CNVs with gene… Read more »
First, thank you to everyone who joined us for our recent webcast, Handling a Variety of CNV Caller Inputs with VarSeq. Also, we would like to thank those that Tweeted #CNVsupport @GoldenHelix or emailed us ([email protected]) to throw in their questions. This was a successful trial run, and we would like to continue engaging with our users through these outlets…. Read more »
Below is a list of highlighted peer-reviewed publications from this month. The Golden Helix team consistently enjoys seeing our software applications hard at work in the field, whether it is clinical whole-exome sequencing, targeted CNV identification, or genotype-phenotype correlations. Enjoy reading below how our suite of VarSeq, VSClinical, and VS-CNV continues to contribute to the progression of genetics research. Looking… Read more »
Advances in high-throughput sequencing have allowed us to be able to detect structural variants such as copy number variants in addition to small variants such as SNVs and indels. We provide users with an industry-leading CNV calling algorithm to detect CNVs directly from their next-generation sequencing data including whole genome, whole exome, and gene panel datasets, and also import CNVs… Read more »
The overall objective of Trio Analysis is to leverage inheritance data to determine the ancestry of variants in the proband, whether that be through transmission from the mother and father, or of de novo origin. We have previously covered family based analysis in this blog post and this webcast, but both of these sources have been based on variants brought… Read more »
If you stay current on the developments of Golden Helix features, you are aware of the substantial evolution of our copy number detection and evaluation capabilities in VarSeq. The process of CNV detection and evaluation is typically handled through the VarSeq graphic user interface. However, in some cases, users benefit from running this process via the command-line interface. Fortunately, Golden… Read more »
In February 2020, the American College of Medical Genetics (ACMG) and the Clinical Genome Resource (ClinGen) published a joint consensus on standards for the interpretation and reporting of copy number variants (CNVs) ranging from large CNVs spanning multiple genes to small intragenic events1. The guidelines consist of over 80 different criteria which are arranged into five distinct sections. These extensive… Read more »
The VarSeq CNV calling algorithm, VS-CNV, is a powerful tool for calling CNVs from the NGS coverage data stored in your BAM files. However, before this algorithm can be deployed in a clinical setting, it must be tuned and validated using data that is representative of your lab’s NGS workflow. In the past, this validation process could be difficult, as… Read more »
Golden Helix has just released VarSeq v2.2.3. In this update, there are notable changes that can improve CNV calling capabilities covered in this webcast. The topics discussed included: Accounting for GC content Improvements to CNV quality flags Using target filtering Updates to the CNV sensitivity and precision settings This blog post will elaborate on these capabilities and demonstrate how they… Read more »
Dr. Auber is the team leader for the molecular genetic diagnosis of hereditary diseases at the Institute for Human Genetics at Hannover Medical School (MHH). MHH is one of the largest hospitals in northern Germany, with one of the largest outpatient clinics for individuals and families dealing with hereditary cancer and predisposition syndromes. Dr. Auber was in high school when… Read more »
In many cases, VarSeq users design their next-gen sequencing workflows for a clinical application. One of the major values of using VarSeq is the standardization of sample analysis via project templates for filtering down to rare variants and isolate any clinically relevant variant. However, VarSeq also doubles as a robust research application as well. There are specific algorithms that can… Read more »
The collaboration between the Clinical Genome Resource (ClinGen) consortium and the American College of Medical Genetics (ACMG) recently developed published guidelines for the interpretation of CNVs called on next-generation sequencing data. These new guidelines are the first to provide a robust set of rules for the interpretation of small intragenic deletions and duplications and are now automated in VSClinical. … Read more »
There is a multitude of interesting new features that have been incorporated into VarSeq 2.2.2. In this blog, I want to continue the discussion of these features and how each can be incorporated into your workflow, and also discuss the application of the Probability Segregation algorithm for copy number variation (CNV) analysis. The Probability Segregation algorithm is a new algorithm… Read more »